Examples of past tools made in the machine shop at Harvard Medical School (prior to May 2015):
Projects
Shaker platform
This large (12" x 24") shaker platform is a classic found in many laboratories around the world. Simply designed, it has proven to be a long-lasting workhorse. It features continuously variable speed control as well as continuous operation. The upper shaking surface is completely covered with 1/8"neoprene foam, and the entire unit is mounted on rubber feet to prevent creeping.
Quick-change perfusion cell
This chamber is designed for microscope experiments that require frequent, rapid changes of cell-plated cover slips. Growing cell colonies can be quickly loaded, examined under the microscope, and then quickly returned to the incubator to maintain viability. The chamber has ample clearance below the cover slip for microscope objectives, allowing oil-immersion observations. The upper section of the chamber can be modified to suit individual perfusion requirements.
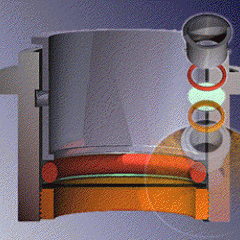
Magnetic separator for iron-bearing cell colonies
This device enables rapid processing in mass cell separation techniques. Extremely powerful magnets positioned on opposite sides of the 50 ml. Falcon© tube quickly attract iron-bearing cells to the inner sidewalls of the tube.These magnets have much greater strength than the magnets normally supplied with commercial separators. Adjustable springs keep the magnets in close contact with the test tube wall. An adapter bushing allows optional insertion of a 15 ml. tube.
Eppendorf© tube freezer rack
This plastic storage rack for Eppendorf test tubes can be used safely for -70 freezer storage. Failure of labeling tape adhesives is a major problem at extremely low temperatures. The inset view in the larger image (click on thumbnail) shows the dovetail slot machined into this rack. Dymo© brand embossed plastic labeling tape (3/8" wide) can be slid into the slot and held captive for secure labeling. The rack holds 150 Eppendorf tubes and is 1 1/4" x 12" x 13".
Bead Arrayer
We have optimized the use of a bead arrayer that immobilizes 384 beads in an equal number of depressions. The arrayer is designed for connection to both a standard vacuum line and a standard nitrogen line. Beads are trapped by application of vacuum to the apparatus, so that excess beads can be brushed away easily and recovered, leaving exactly 384 beads on the arrayer in a regular 24 X 16 matrix. To deposit individual beads into the wells of a microtiter plate, the arrayer is inverted onto the plate, such that one bead is suspended over each well of the plate, and the vacuum relieved by pressurization with nitrogen. Beads are thus pushed out of the depressions and down into the wells of the microtiter plate.